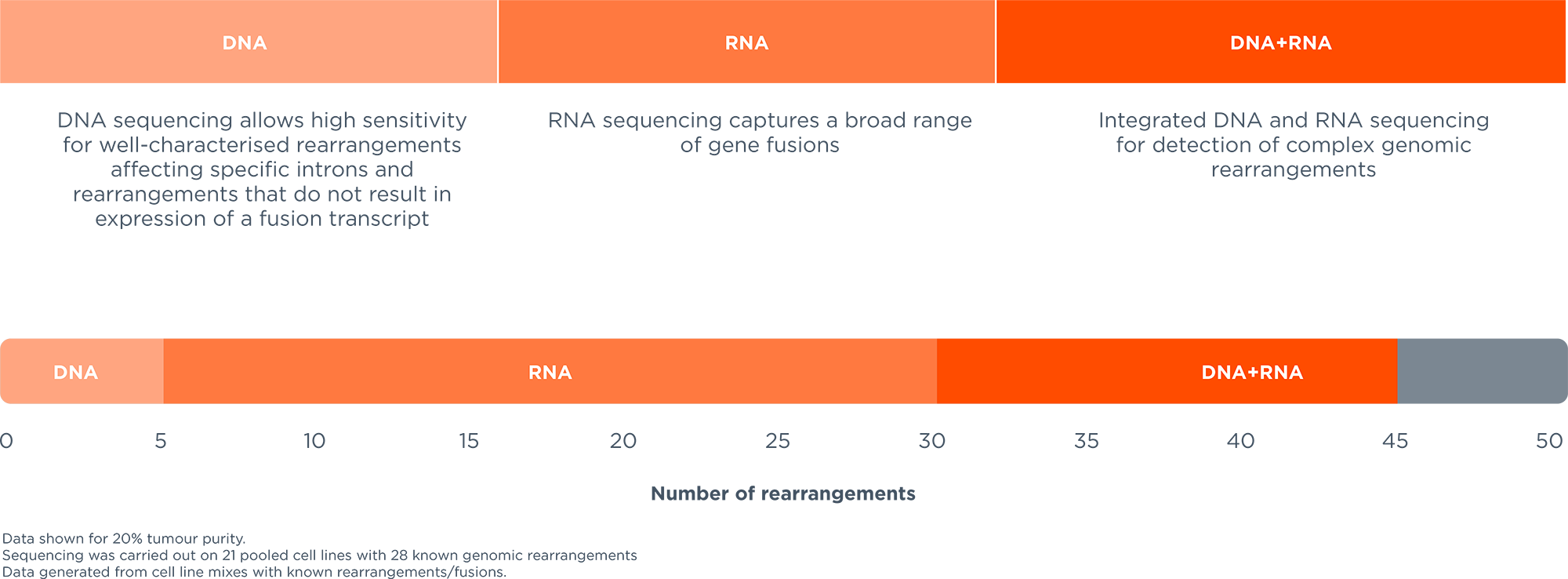
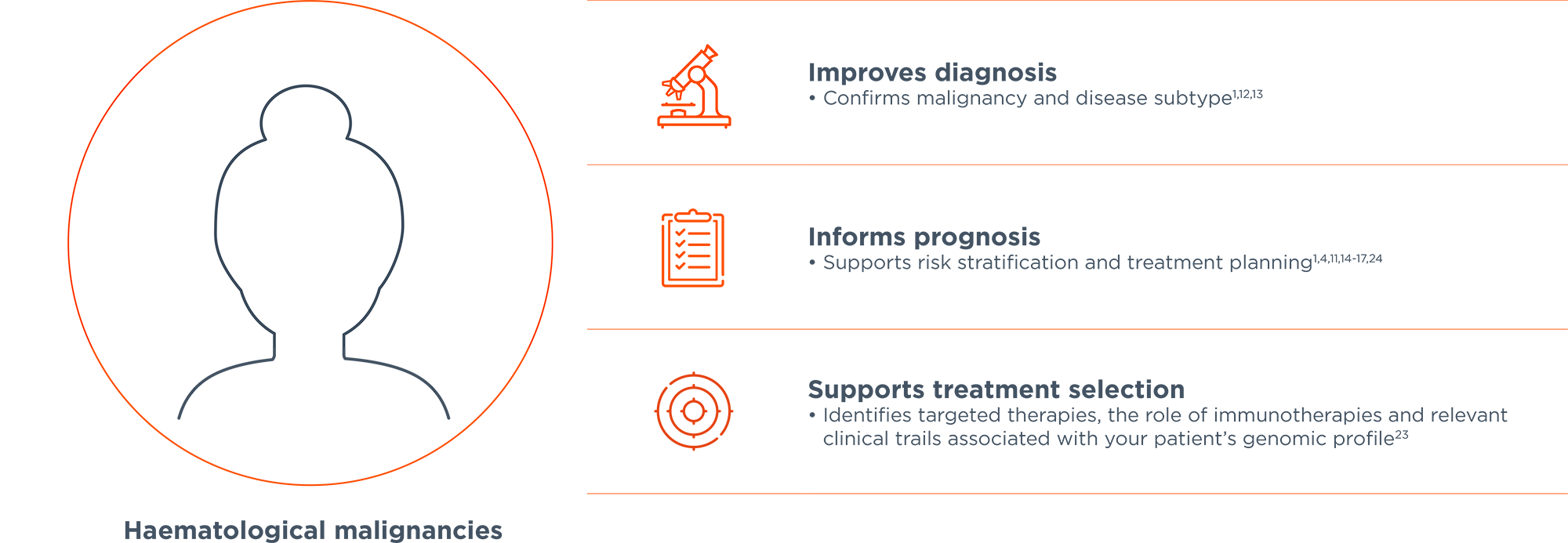
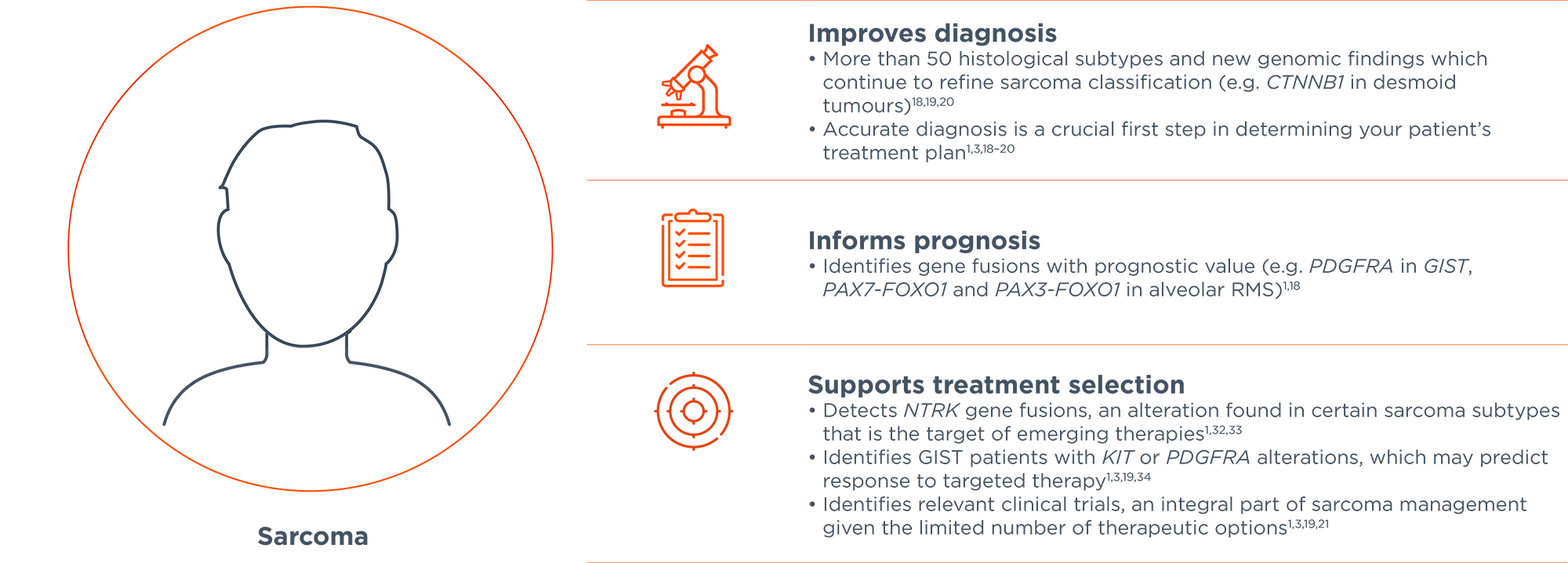
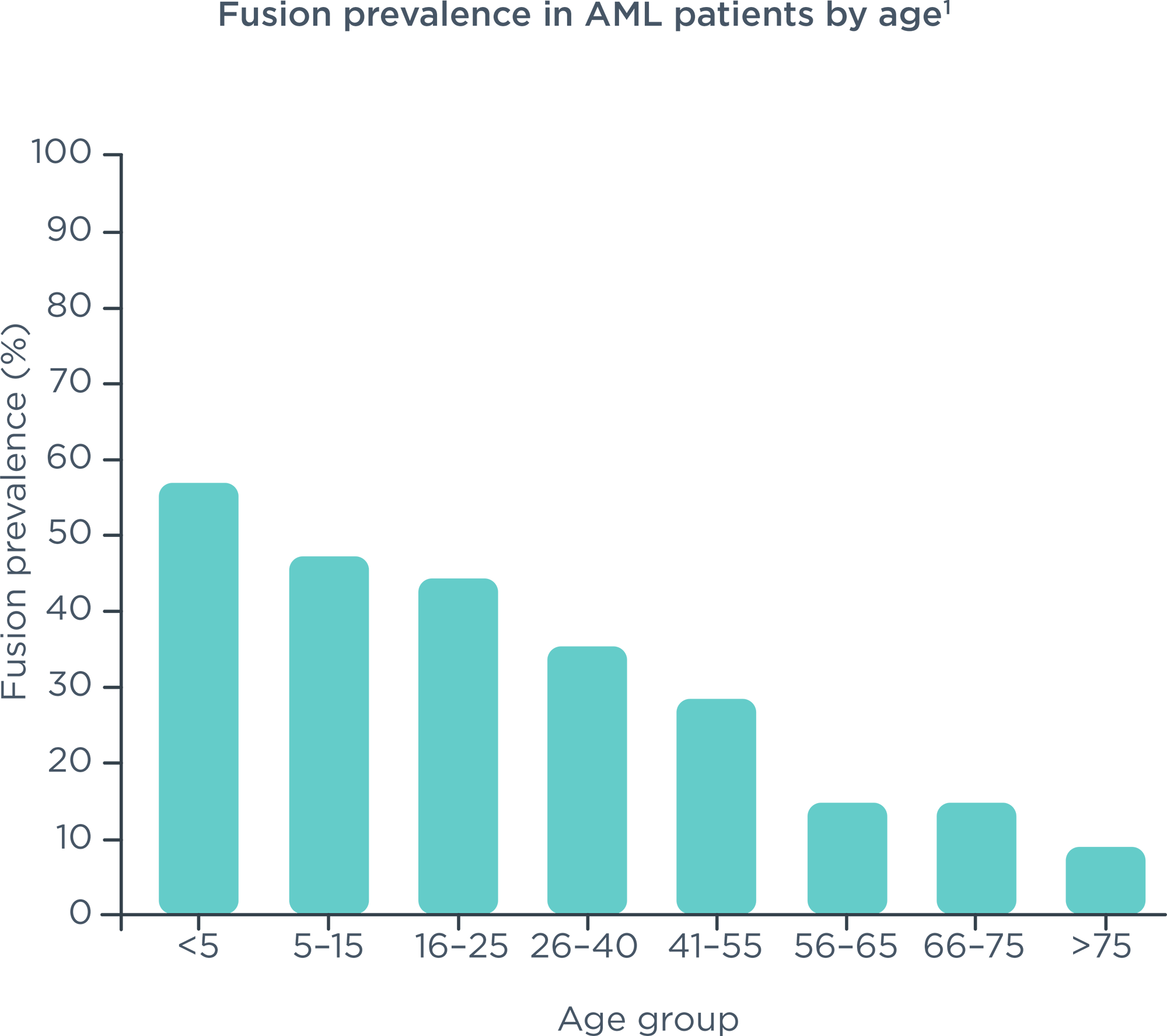
Comprehensive genomic profiling service for haematological malignancies and sarcomas; to guide diagnosis, prognosis and treatment selection and personalise patients' treatment plans.1-3
This website is a global information resource. It is intended for healthcare professionals only outside of the United States of America (US) who are interested in information on Foundation Medicine®. This site is not intended to provide medical advice and/or treatment guidance. If you are a US healthcare professional click here.
This site is produced by Roche as a partner of Roche Foundation Medicine.